- Therapeutic Cataract & Refractive
- Lens Technology
- Glasses
- Ptosis
- Comprehensive Eye Exams
- AMD
- COVID-19
- DME
- Ocular Surface Disease
- Optic Relief
- Geographic Atrophy
- Cornea
- Conjunctivitis
- LASIK
- Myopia
- Presbyopia
- Allergy
- Nutrition
- Pediatrics
- Retina
- Cataract
- Contact Lenses
- Lid and Lash
- Dry Eye
- Glaucoma
- Refractive Surgery
- Comanagement
- Blepharitis
- OCT
- Patient Care
- Diabetic Eye Disease
- Technology
Lumenis introduces OptiLight at Vision Expo West
The new intense pulsed light technology targets meibomian gland dysfunction
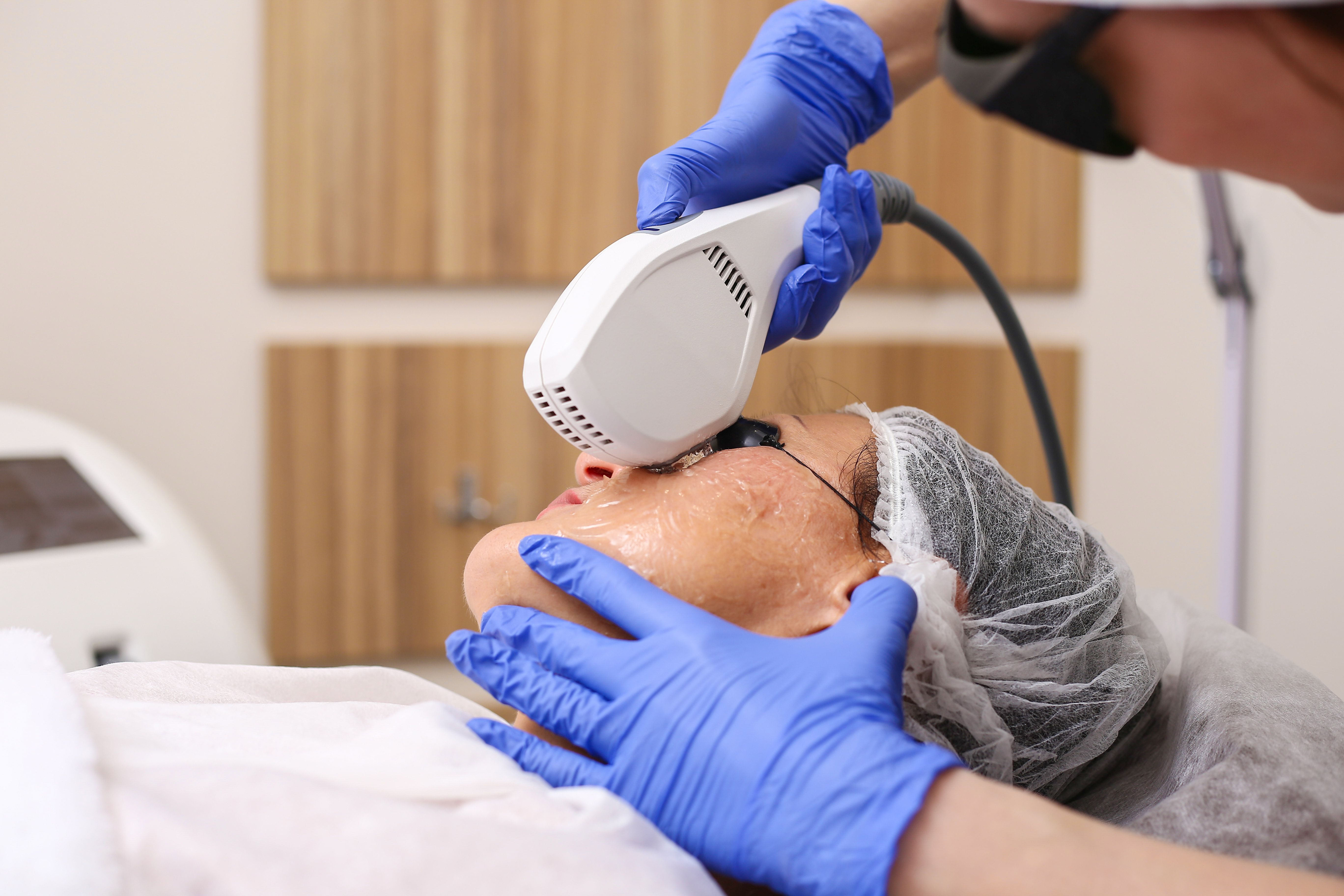
Lumenis launched its OptiLight intense pulsed light (IPL) device at Vision Expo West 2021 in Las Vegas, Nevada.
The FDA granted the company authorization for OptiLight, which is designed to improve dry eye disease through the use of IPL.
Study results
A multicenter, double-blinded, randomized controlled trial revealed that the Lumenis IPL system with Optimal Pulse Technology (OPT) significantly improved tear break-up time, meibum quality, and meibomian gland expressibility.1
Related: Optimizing management of dry eye disease
“Unless we address the underlying inflammation, it’s difficult to meaningfully impact dry eye disease,” said Steven J. Dell, lead investigator in the clinical trial, in a statement.1 “OptiLight helps us to address the inflammation, and improves signs of dry eye disease due to MGD, positioning it as a valuable tool in our dry eye toolkit.”
Related news
In related news, private equity firm Baring Private Equity Asia and Lumenis have partnered to accelerate the investment in vision and aesthetics vertical markets following the sale of Lumenis Surgical for $1.07B.
Lumenis recently completed the sale of its surgical division to Boston Scientific Corporation, a global medical device manufacturer.2
The company plans to increase its vision and aesthetic businesses through its research, development, and marketing efforts.2
Related: How to implement IPL into an optometry practice
Reference
1. Britton G. Lumenis receives FDA approval for Its IPL device to manage dry eye disease and launches OptiLight. Lumenis news release. April 29, 2021. Accessed September 22, 2021. https://lumenis.com/medical/news/lumenis-receives-fda-approval-for-its-ipl-device-to-manage-dry-eye-disease-and-launches-optilight/
2. Lumenis and Baring Private Equity Asia to accelerate investment in aesthetics and vision verticals following the strategic sale of Lumenis Surgical for $1.07B. Lumenis news release. September 1, 2021. Accessed September 22, 2021. https://lumenis.com/medical/news/lumenis-and-baring-private-equity-asia-to-accelerate-investment-in-aesthetics-and-vision-verticals-following-the-strategic-sale-of-lumenis-surgical-for-1-07b/
