A chance to be the hero
Reducing the burden of drops can improve patients’ QOL and prevent progression of glaucoma.


The ultimate goal of glaucoma treatment is to keep our patients’ seeing for the rest of their lives. Most of us remember very clearly the first time we saw a patient go blind from glaucoma—a terrible outcome we would like to avoid.
Topical IOP-lowering medications, and particularly the prostaglandin analogue (PGA) class typically used as first-line therapy, have been essential in preserving vision for millions of Americans. However, as with any medication, there are some disadvantages to IOP-lowering drops.
We have all had patients who have been unresponsive to IOP-lowering medications only to find out that they were placing the drops in their ears or mouth, or, more commonly, were just missing their eye.
It is no surprise that findings from multiple studies confirm that patients are doing a much worse job of it on their own.1,2
Daily use of medications has a psychological impact, because it reminds patients that they have an incurable disease, and a financial impact.
In one study, nearly 40% of patients were prescribed 2 or more topical medications at 60 months, while just over 9% of participants were prescribed 3 or more medications.3
Further, an additional study included 75% of patients with mild to moderate glaucoma requiring 2 or more prescription eye drops to control pressure.4 Most of these patients are spending money out of pocket every month to fill those prescriptions.
The chronic, long-term use of any topical therapy has adverse effects on the ocular surface. Approximately half of glaucoma patients have abnormal Ocular Surface Disease Index (OSDI) scores.5
Studies have shown that a vast majority (92%) of those using a PGA drop for more than 1 year have some level of meibomian gland disease (MGD).6
Although there may be some aspects of the active ingredients that contribute to ocular surface disease (OSD), the greatest impact likely comes from the chronic use of preservatives. Studies have confirmed that the rate of OSD rises with the number of topical medications being used by glaucoma patients.7
We know that OSD has a negative effect on patients’ quality of life (QOL), but even beyond that, it can affect their adherence with glaucoma therapy.
Lid margin damage and changes in the meibomian gland structure or function are significant factors in reduced adherence with glaucoma medications because as patients start to become symptomatic they are less likely to use their medications.8
Patients with ocular surface adverse effects from glaucoma medications have been shown to have a more rapid progression of visual field loss.9 It is not hard to imagine why this is the case.
If a patient’s eyes are red or they experience burning and stinging when instilling drops, they may take a day or two off to avoid dealing with the irritation. That break stretches to a week or more, or maybe they stop using drops entirely until the week before an eye care appointment.
When patients intermittently use their glaucoma medications, fluctuation in IOP occurs, leading to a higher risk of progression. Topical IOP-lowering medications are not going away, nor should they. They are essential in the management of our glaucoma patients, but are there opportunities to reduce medication for our patients?
Cataracts bring opportunity
As our patients with glaucoma develop cataracts, we have a unique opportunity to reduce all these unwanted adverse effects by making them less dependent on topical drops to maintain an acceptable IOP.
Clinical trials have shown that cataract surgery itself will lower IOP. However, combining cataract surgery with a minimally invasive glaucoma surgery (MIGS) procedure has an even better chance of reducing IOP and medication usage than cataract surgery alone, without any additional surgical risk.8
In a study conducted by one of us (Schweitzer), patients with cataracts and glaucoma undergoing cataract surgery with implantation of 1 or 2 trabecular microbypass stents (iStent or iStent inject; Glaukos) medications were reduced by 60% (from 1.5 to 0.6 medications). We also saw a significant corresponding improvement in ocular surface health within only 3 months of surgery.9
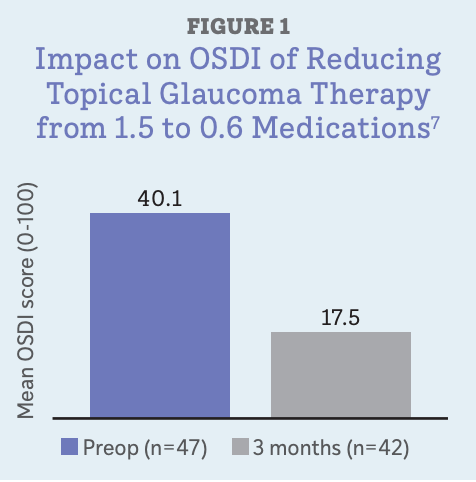
With no other ocular surface interventions, simply reducing the number of topical medications moved patients’ mean OSDI score from severe to mild (Figure 1) and brought nearly 60% of them into the “normal” range for OSDI, compared with less than 10% preoperatively. Staining was significantly reduced from baseline, and tear film breakup time was 49% longer as well.
Quality-of-life impact
In a recent pos thoc analysis of patient-reported outcomes (PROs) from the iStent inject pivotal trial (NCT01461291), investigators again observed a positive impact on OSDI scores, along with meaningful improvements in vision-related QOL among patients who underwent trabecular microbypass surgery.10
In this study, nearly 2000 patients were randomized 3:1 to cataract surgery with the 2 trabecular microbypass stents (n = 1490) or cataract surgery alone (n = 460).11
PROs were measured using the OSDI and the 25-item version of the National Eye Institute Visual Function Questionnaire (NEI-VFQ-25), a widely used vision-related QOL instrument that measures the impact of symptoms on a range of factors, including general vision, near and distance activities, mental health, social functioning, and driving. The OSDI and VFQ-25 responses were collected at baseline and throughout the 2-year follow-up.
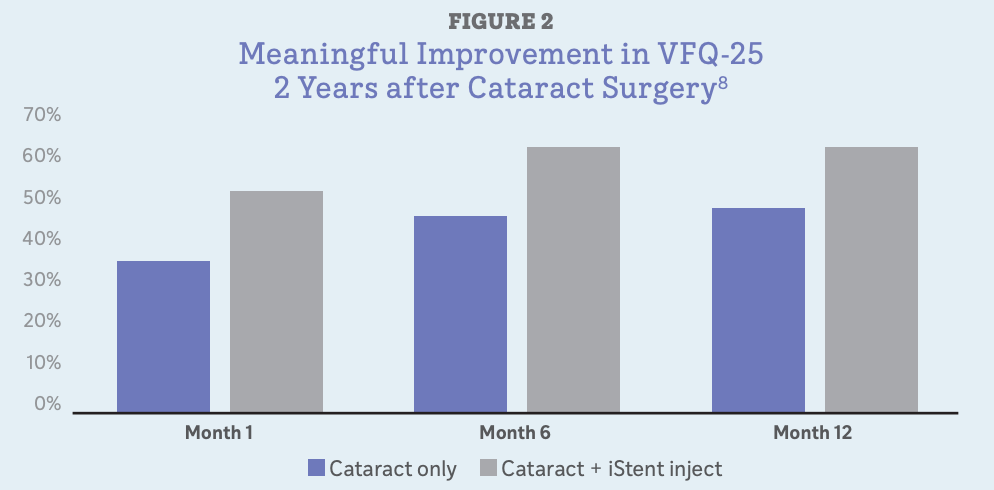
Meaningful VFQ-25 improvements were observed in both groups at all time intervals, with greater improvements noted in the cataract surgery plus MIGS group (Figure 2). Differences in the VFQ-25 response rates were most pronounced in the subscales for general vision, ocular pain, and driving.12
Across both groups (cataract surgery only and cataract + MIGS), those patients who were medication free at 24 months were most likely to have achieved meaningful gains on the VFQ-25.
In other words, decreasing their medication burden—something that was more likely to happen in the patients undergoing cataract plus MIGS than cataract surgery alone—improved QOL.
Although we have long suspected that MIGS procedures in general could affect vision-related QOL, this study is actually the first of its kind to show such an improvement. The lesson we can take from this research is that recommending a MIGS procedure at the time of cataract surgery offers a real opportunity to help our patients with open-angle glaucoma.
Although this is particularly important for patients who are poorly controlled on multiple medications, we would argue that it is worthwhile even among those who are well controlled and reasonably adherent.
As these patients age, they may experience increased difficulty instilling drops, due to arthritis or cognitive decline. Furthermore, preserving the ocular surface should be a major factor that is considered when managing these patients.
It is important for optometrists not only to be proactive in recommending MIGS at the time of surgery, but also to be able to follow these patients postoperatively, manage glaucoma,and decrease medication burden where possible.
References:
1. Okeke CO, Quigley HA, Jampel HD, et al. Adherence with topical glaucoma medication monitored electronically: the Travatan Dosing Aid study.Ophthalmology. 2009;116(2):191-199. doi:10.1016/j.ophtha.2008.09.004
2. Nordstrom BL, Friedman DS, Mozaffari E, Quigley HA, Walker AM. Persistence and adherence with topical glaucoma therapy.Am J Ophthalmol. 2005:140(4):598-606. doi:10.1016/j.ajo.2005.04.051
3. Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol 2002;120:701–713.
4. Lichter PR, Musch DC, Gillespie BW, Guire KE, Janz NK, Wren PA, Mills RP; CIGTS Study Group. Interim clinical outcomes in the Collaborative Initial Glaucoma Treatment Study comparing initial treatment randomized to medications or surgery. Ophthalmology. 2001 Nov;108(11):1943-53.
5. Fechtner RD, Godfrey DG, Budenz D, Stewart JA, Stewart WC, Jasek MC. Prevalence of ocular surface complaints in patients with glaucoma using topical intraocular pressure-lowering medications. Cornea. 2010;29(6):618-621. doi:10.1097/ICO.0b013e3181c325b2
6. Mocan MC, Uzunosmanoglu E, Kocabeyoglu, et al. The association of chronic topical prostaglandin analog use with meibomian gland dysfunction. J Glaucoma. 2016;25(9):770-774. doi:10.1097/IJG.0000000000000495
7. Rossi GCM, Pasinetti GM, Scudeller L, Raimondi M, Lanteri S, Bianchi PE. Risk factors to develop ocular surface disease in treated glaucoma or ocular hypertension patients. Eur J Ophthalmol. 2013;23(3):296-302.
8. Lee TH, Sung MS, Heo H, Park SW. Association between meibomian gland dysfunction and compliance of topical prostaglandin analogs in patients with normal tension glaucoma. PLoS One. 2018;13(1):e0191398.
9. Denis P. Adverse effects, adherence and cost-benefits in glaucoma treatment. Eur Ophthalmic Rev. 2011;5(2):116-122. doi:10.17925/EOR.2011.05.02.116
10. Samuelson TW, Sarkisian SR Jr, Lubeck DM, et al; iStent inject Study Group. Prospective, randomized, controlled pivotal trial of an ab interno implanted trabecular micro-bypass in primary open-angle glaucoma and cataract: two-year results. Ophthalmology. 2019;126(6):811-821.
11. Schweitzer JA, Hauser WH, Ibach M, et al. Prospective Interventional Cohort Study of Ocular Surface Disease Changes in Eyes After Trabecular Micro-Bypass Stent(s) Implantation (iStent or iStent inject) with Phacoemulsification. Ophthalmol Ther. 2020;9(4):941-953.
12. Samuelson TW, Singh IP, Williamson BK, et al. Quality of life in primary open-angle glaucoma and cataract: an analysis of VFQ-25 and OSDI from the iStent inject pivotal trial. Am J Ophthalmol. 2021;229:220-229. doi:10.1016/j.ajo.2021.03.007

Newsletter
Want more insights like this? Subscribe to Optometry Times and get clinical pearls and practice tips delivered straight to your inbox.